|
Among this multitude of functions, the ability of chaperones to mediate the assembly of oligomeric complexes is of particular interest as advances in functional proteomics have revealed that a significant portion of a cell's proteome realize their functional potential in multiprotein complexes rather than as singular entities ( Sali et al. The heat shock proteins (HSPs), among other ubiquitous chaperone families, have well-documented roles in preventing the aberrant association or aggregation of proteins in addition to facilitating protein synthesis, translocation, de novo folding, and higher ordered assembly of multiprotein complexes ( Hendrick & Hartl 1993, Neuer et al. Originally identified as inducible proteins involved in the protection of cells from multiple stresses, molecular chaperones are now recognized as participants in a diverse range of functions due to their ability to selectively bind to hydrophobic residues of target proteins, directing their involvement in correct protein folding or degradation pathways ( Ellis 1987, Hendrick & Hartl 1993). Further analyses of the intermediary proteins that facilitate the expression of key players in sperm–egg fusion are likely to deliver important insights into this unique event, which culminates in the cytoplasmic continuity of the male and female gametes. Despite these findings, the analysis of oocyte surface multiprotein complexes is currently lacking. Preliminary studies of the oocyte plasma membrane have also revealed the presence of lipid rafts comprising several molecular chaperones, raising the possibility that similar mechanisms may be involved in the activation of maternal fusion machinery and the regulation of oocyte plasma membrane integrity. While the activation of these molecular chaperones and the mechanisms by which they shuttle proteins to the surface of the cell remain the subject of ongoing investigation, a compelling suggestion is that these processes are augmented by dynamic membrane microdomains or lipid rafts that migrate to the apical region of the sperm head after capacitation. Novel research suggests that these highly specific cellular interactions are facilitated by multiprotein complexes that are delivered to and/or assembled on the surface of the gametes by molecular chaperones in preparation for sperm–egg interaction. Step by step, we were able to achieve our results.The remarkable complexity of the molecular events governing adhesion and fusion of the male and female gametes is becoming apparent. This led to hypotheses about the ribosome interaction, which we have studied using further genetic, biochemical and kinetic approaches. Computer simulations of the molecular dynamics of Ssb provided important indications of molecular interactions within the Ssb protein. She further comments on the Ssb-chaperone results: "It was great team work within the University of Konstanz and with renowned teams from Heidelberg and Stanford. "How the RAC-Hsp70 system operates in higher cells and what effects it has on proteins relevant for diseases will be a huge topic in the SFB," says Deuerling. This is why the researchers assume that the RAC-Hsp70-chaperone system is generally very important for correct folding of proteins and for keeping the cells fit. In higher cells, RAC probably cooperates with other Hsp70 chaperones. In contrast to the co-chaperone RAC, the ribosome-bound chaperone Ssb can only be found in fungi such as yeast. Finding this exact position at the ribosome is of crucial importance for the effectiveness of the chaperone. "We believe that these multivalent interactions with direct attachment sites and the cooperation with RAC allow the chaperone Ssb to position itself at the ribosome in an optimal way," says Deuerling. However, if co-chaperone RAC is absent, abnormal protein folding occurs in Ssb mutant cells, resulting in pronounced cellular defects. 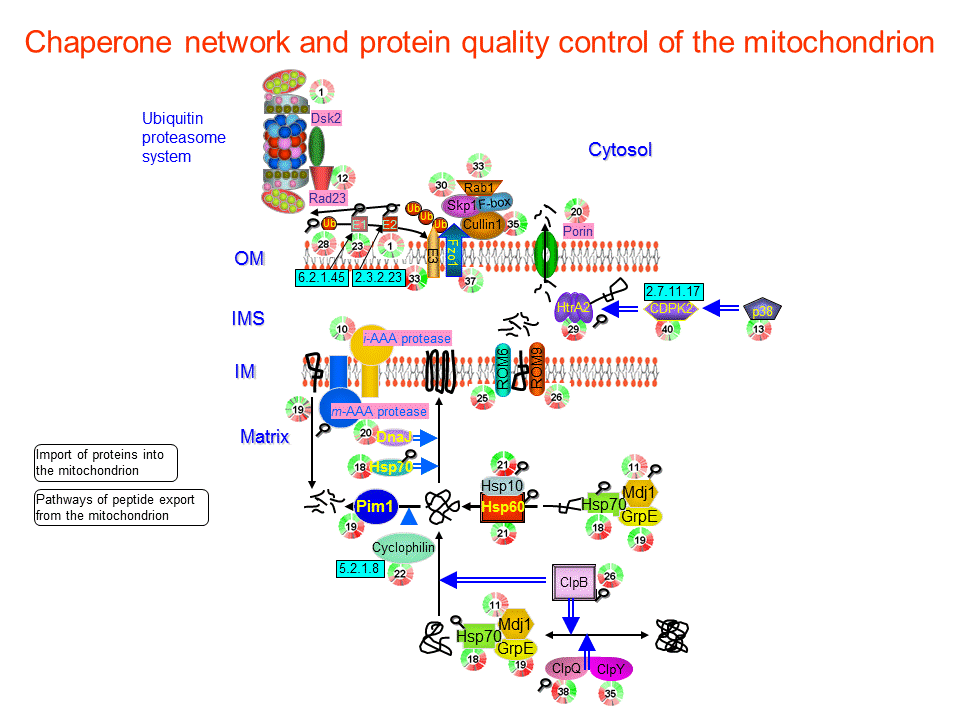
If they are mutated, nothing changes for the yeast cells. In several experiments, the biologists found out that the Ssb attachment sites are not essential under normal laboratory conditions. 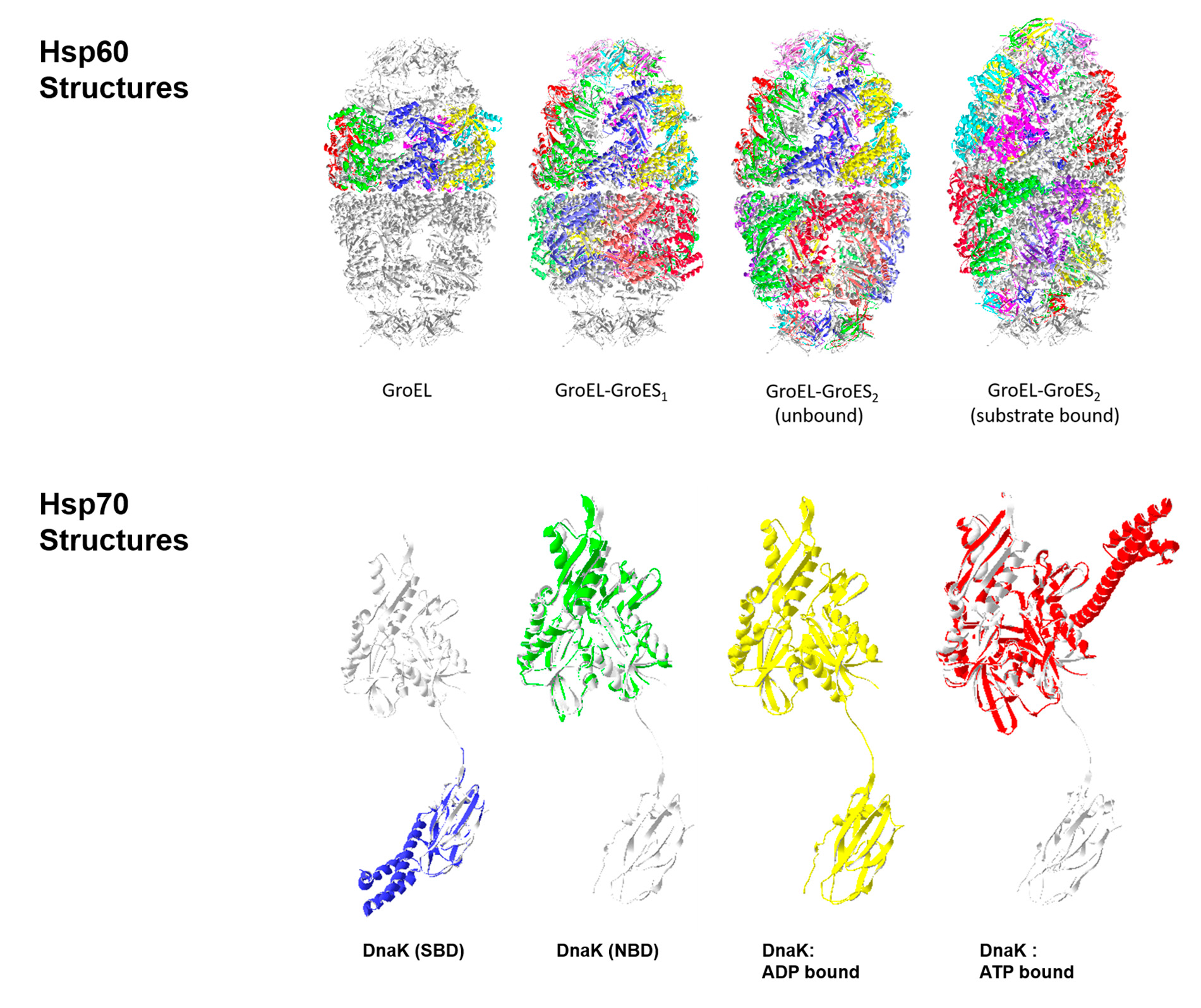
Anne Hanebuth significantly contributed to the discovery of the Ssb attachment sites in her doctoral thesis. Only a few amino acids provide Ssb with this additional feature and show how flexible Hsp70 chaperones can be," explains molecular biologist Elke Deuerling. "This ability is unique and cannot be detected at first glance when comparing Ssb with other Hsp70 chaperones. It is active at a very early stage, when new proteins are being synthesised in the ribosome. Ssb is the only family member that is in direct contact with the ribosome. Hsp70 chaperones play a central role for preventing abnormal protein folding as well as for the transport of proteins. The chaperone Ssb is a member of the highly conserved Hsp70 chaperone family found in all living organisms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
